Executive Summary
This review describes the evidence linking birth outcomes to risk of developing diabetes mellitus (DM). DM prevalence is increasing unacceptably, especially in low- and middle-income countries (LMICs), placing a heavy burden of morbidity on individuals and driving higher health care costs and lower productivity.
This review was conducted as part of SPRING's work on the Early-Life Nutrition Linkages to Noncommunicable Disease (ELN-NCD) simulation model. The ELN-NCD simulation model examines how birth outcomes relate to later life risk of nutrition-related NCDs. The final model focuses only on cardiovascular disease, but during the systematic evidence review, a substantial body of literature was found on linkages between birth outcomes and DM.
Twenty-three articles were reviewed and are analyzed here. A majority of studies found increased risk of DM and/or pre-DM conditions (fasting glucose, fasting insulin) with lower birthweight (BW), and some studies found a U-shaped relationship, meaning that at a certain BW, the risk of DM begins to increase again. SPRING’s earlier ELN-NCD work models mortality and morbidity from later life cardiovascular disease associated with early-life undernutrition. In the case of DM, it is more difficult to make such estimations of reductions in mortality and morbidity. While it appears that the majority of the studies found a significant inverse relationship between BW (below a certain threshold) and DM risk, there is variance in the nature of the relationship, how persistent it is throughout the life cycle, and what modifies the risk. Also, the relationship is difficult relationship to understand because DM has a multifactorial and dynamic etiology, including strong contributions from childhood and adult body weight trajectory, lifestyle, and genetic factors.
Consensus is growing that BW does have significant effect on DM risk, but more work is needed to conclusively define that association in LMICs, in different ethnic groups, and across the spectrum of BW. Efforts to reduce low birthweight (LBW) should continue, given its proven relationship with neonatal mortality; this review’s attempt to improve estimates of the relationship between LBW and later life NCD risk should be considered further support to bolster the case for efforts to reduce LBW.
Introduction
This review describes the evidence linking birth outcomes to risk of developing diabetes mellitus (DM, or Type 2 diabetes). Globally, prevalence of DM among adults (aged 20–79 years) was estimated to be about six percent in 2010, affecting 285 million adults (Shaw, Sicree, and Zimmet 2010). This is expected to increase to just under eight percent (439 million adults) by 2030, with the majority of the growth in low- and middle-income countries (LMICs). There is general consensus that the rising tide of DM is unacceptable: the disease places a heavy burden of morbidity on individuals and is one of the major drivers of health care costs and lost productivity (The Lancet 2014).
While relatively few individuals with DM will die directly from an imbalance in glucose or other metabolic imbalances (Zargar et al. 1999), the disease is a risk factor for more terminal conditions, such as stroke, heart failure, and renal failure (Peters, Huxley, and Woodward 2014; Holman, Sourij, and Califf 2014; Perneger et al. 1994; Yoon et al. 2006). DM is also an identified risk factor for several types of cancer, though the mechanism by which it increases risk is not entirely understood (Shi and Hu 2014). This is further complicated by the differentials in risk for early- and late-onset DM (S.H. Song and Hardisty 2008).
There is a progression of conditions that can lead to onset of DM, including obesity, insulin resistance, and impaired glucose tolerance. The general constellation of pre-DM conditions can be roughly mapped (Figure 1); however, this process is not linear, and an individual can manifest any of these conditions out of this sequence, or stop progression prior to onset of DM.
Figure 1. Potential Pathways to DM from Pre-DM Conditions
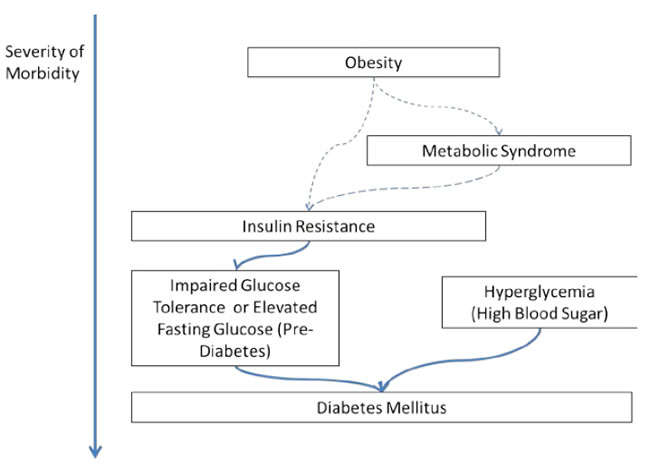
One can develop DM from any stage in Figure 1, though the risk appears to be higher as these symptoms progress (Sung et al. 2012). The focus of efforts to prevent DM and pre-DM conditions has been on adolescent or adult interventions to improve diet and increase exercise (Ley et al. 2014); however, in other areas of research, notably obesity reduction, few studies have been able to show sustained, long-term improvements due to improved diet and exercise regimes (Monasta et al. 2011; Ho et al. 2013; Curioni and Lourenço 2005). Many more studies have devoted time to treatment options for DM via pharmacology (e.g., Li et al. 2010).
More recently, a small body of evidence has emerged around reduction of DM risk via improving birth outcomes, as part of the life course hypothesis that an individual’s metabolism is affected by conditions in utero (Godfrey, Gluckman, and Hanson 2010). Hattersley and Tooke (1999) coined the term “fetal insulin hypothesis.” Birth outcomes such as LBW, preterm birth, or small for gestational age have been linked to adult DM, either directly or through prediabetic conditions at various stages of infancy, adolescence, and adulthood (e.g. Kaijser et al. 2009; Whincup et al. 2008; Tinnion et al. 2013).
This review will summarize the current state of evidence on the associations between birth outcomes and DM or pre-DM conditions (such as insulin resistance or impaired glucose tolerance). It will also provide information on some of the primary post-birth modifiers of DM risk.
Methods
This review was conducted as part of SPRING's work on the Early-Life Nutrition Linkages to Noncommunicable Disease (ELN-NCD) simulation model. The ELN-NCD simulation model examines how birth outcomes relate to later life risk of nutrition-related NCDs. The final model focuses only on cardiovascular disease, but during the systematic evidence review, a substantial body of literature was found on linkages between birth outcomes to DM.
Evidence was collected via a systematic stand-alone review of all evidence related to the “hub” of birth outcomes and impact on risk of DM or pre-DM conditions. The focus was specifically on BW, not small for gestational age or preterm birth; however, if an article examined one of these conditions in addition to LBW, it was included.
The primary pre-DM conditions used in the literature were:
- insulin resistance (IR)
- impaired glucose tolerance (IGT)
- elevated fasting glucose
- elevated fasting insulin
- metabolic syndrome1
The search was conducted via Google Scholar for English-language articles from the past 20 years that included the search terms listed in the Annex. Articles from LMIC countries were preferred, but no study was excluded if it was from a high-income country. After all terms were exhausted, articles were ranked based on quality and location. If a study had a particularly small sample size (<100); a lack of statistical rigor (no control group or lacked tests of significant difference); or shortcomings in reporting of results (did not report sufficient information to glean risk ratios, or raised concern on the design of the study), it was excluded from the final review tables.
Extraction of information was done for all eligible studies and formed the basis for the tables in this review.
Results of Review
Twenty-five articles were found that met the search criteria. Four of the 27 articles found were excluded due to sample size or reporting issues (Sugihara et al. 2008; Choi et al. 2000; Anazawa, Atsumi, and Matsuoka 2003; Yajnik 2002). Other relevant reviews using gestational age as an outcome were found, but they were excluded from this review as they did not fit the search criteria of using BW as the hub for the risk estimation. (See Parkinson et al. 2013; Tinnion et al. 2013; Darendeliler et al. 2008 for more information on these alternative measures of birth outcomes).
Another set of literature focused on large for gestational age or high BW, a condition often caused by gestational diabetes (Boney et al. 2005; Carpenter 2007). Due to the frequent complication of gestational diabetes, these studies were not summarized here for equal comparison against the evidence for LBW; however, they were included if the primary purpose of that study was to review BW or LBW.
In the remaining 23 studies found in the review, the following measures of BW outcomes were used:
- Continuous measure of BW
- Categorical measure of BW (groupings of kg or g measures)
- A binary measure of BW (LBW, <2,500g)/normal BW)
Four of the included studies were systematic reviews of the topic and pull from a larger population for their risk estimates (three of the four included some form of meta-analysis). The rest of the studies employed a wide range of methods to develop their estimates, including retrospective chart review, longitudinal pre- and post- designs, as well as prospective birth cohort studies.
Relationship with Risk
A majority of studies found increased risk of DM and/or pre-DM conditions (fasting glucose, fasting insulin) with lower BW (see Table 1). Of the 23 studies, 11 found unambiguous inverse associations with BW; that is, as BW increased, risk of developing a DM or a pre-DM condition appeared to go down. An additional four studies found an inverse relationship when adult weight, waist circumference, or BMI were controlled for. Four more studies saw an inverse association with insulin resistance, but they documented non-significant associations with other pre-DM conditions (Fall et al. [1998] only saw this association in men). This association appeared more often when the studies’ focus was on DM outcomes (top of Table 1), less so with pre-DM conditions (bottom of Table 1). However, few studies reported whether they tested for anything other than a linear trend, which imposes a heavy assumption on the shape of the trend over the full range of BW.
Table 1. Relationship between BW and DM or Pre-DM Condition
| # | Author-Date | Birth Outcome | Outcome Type | Location | Diabetes Outcome | Relationship |
|---|---|---|---|---|---|---|
| Legend | - = inverse association. As birthweight goes up, risk of adverse event goes down. | 0= No statistically significant association found. | + = direct association. As birthweight goes up, risk of adverse event goes up. | |||
| 1 | Bonsdorff et al. 2013 | BW (g) | (categorical) | Iceland | DM | - |
| 2 | Curhan et al. 1996 | BW (lbs) | (categorical) | USA | DM | - |
| 3 | Fall et al. 1998 | BW (kg) | (continuous) | India | DM | 0 |
| 4 | Harder et al. 2007 | BW (g) | (categorical) | Review; Various | DM | U-shaped |
| 5 | Kaijser et al. 2009 | BW (g) | (continuous) | Sweden | DM | + |
| 6 | Newsome et al. 2003 | BW (multi) | multi | Review; Various | DM | - (after adjusting for current weight) |
| 7 | Norris et al. 2012 | BW (kg) | (continuous) | Brazil, Guatemala, India, the Philippines, and South Africa | DM/fasting glucose | - |
| 8 | Phipps et al. 1993 | BW (kg) | (categorical) | UK | DM | - |
| 9 | Raghupathy et al. 2010 | BW (z-score) | (continuous) | India | DM/ IGT | - |
| 10 | Song et al. 2012 | BW (g) | (categorical) | Review; Various | DM/ IGT | U-shaped |
| 11 | Tian et al. 2006 | BW (g) | (categorical) | China | DM | - |
| 12 | Wei et al. 2003 | BW (g) | (categorical) | Taiwan | DM | U-shaped |
| 13 | Whincup et al. 2008 | BW (kg) | (continuous) | Review; Various | DM | - |
| 14 | Bavdekar et al. 1999 | BW (kg) | (continuous) | India | fasting insulin/ IR, fasting glucose | -, 0 |
| 15 | Bhargava et al. 2004 | BW (kg) | (continuous) | India | IGT, fasting insulin, IR | 0,-,- |
| 16 | Bonsdorff et al. 2013 | BW (g) | (categorical) | Iceland | IR, fasting glucose, DM | - |
| 17 | Fall et al. 1995 | BW (lbs) | (continuous) | UK | fasting glucose | - |
| 18 | Fall et al. 1998 | BW (kg) | (continuous) | India | IR | - (men), 0 (women) |
| 19 | Frontini et al. 2004 | LBW/NBW (g) | (binary) | USA | fasting glucose, IR | 0,- |
| 20 | Hovi et al. 2007 | VLBW (g) | (binary) | USA | IR, IGT | - , - |
| 21 | Mi et al. 2000 | BW (g) | (continuous) | China | fasting glucose | - |
| 6 | Newsome et al. 2003 | BW (multi) | multi | Review; Various | IR | - (after adjusting for current weight) |
| 7 | Norris et al. 2012 | BW (kg) | (continuous) | Brazil, Guatemala, India, the Philippines, and South Africa | IR | - (after controlling for Adult waist circumference) |
| 9 | Raghupathy et al. 2010 | BW (z-score) | (continuous) | India | IR | - (after controlling for Adult BMI) |
| 22 | Stein et al. 2002 | BW (kg) | (continuous) | Guatemala | fasting glucose | 0 |
| 23 | Stewart et al. 2010 | BW (kg) | (continuous) | Nepal | IR & fasting glucose | 0, 0 |
| 15 | Curhan et al. 1996 | BW (lbs) | (categorical) | USA | diabetes mellitus | - |
| 16 | Fall et al. 1998 | BW (kg) | (continuous) | India | diabetes mellitus | 0 |
| 5 | Harder et al. 2007 | BW (g) | (categorical) | Review; Various | diabetes mellitus | U-shaped |
| 7 | Kaijser et al. 2009 | BW (g) | (continuous) | Sweden | diabetes mellitus | + |
| 17 | Newsome et al. 2003 | BW (multi) | multi | Review; Various | diabetes mellitus | - (after adjusting for current weight) |
In fact, when looking at the four literature reviews included in this review, two of the studies found evidence of a nonlinear, U-shaped relationship. This means that at a certain BW, the risk of DM plateaus and begins to increase again (Harder et al. 2007; Y. Song et al. 2012). The remaining two, both meta-analyses, found an inverse relationship overall but mentioned a sub-section of studies that had U-shaped relationships (Newsome et al. 2003; Whincup et al. 2008). Whincup et al. (2008) in particular noted that U-shaped results occurred in the three Native American population studies included in the review; they were excluded in the final meta-analysis to reduce heterogeneity.
There are two primary explanations posited for this mix of findings.
- Differences in starting BW in different levels of economic prosperity, shifting the distribution of children falling into the low, normal, and high BW categories. In LMICs, a greater proportion of babies will fall in the LBW category, while in high-income or food-secure countries, the distribution will include more high BW babies. Indeed, two of the studies supporting a U-shaped relationship (Harder et al. 2007; Y. Song et al. 2012) were reviews that included several high-income country studies; a third single study (Wei et al. 2003) also found a U-shaped association in a Taiwanese population.
- Differences in risk for different ethnicities. Some evidence showed that certain ethnicities are at greater risk of gestational diabetes and other maternal metabolic imbalances that could lead to macrosomia (babies born at or above 4,000g). These imbalances also carry an independent risk for metabolic disorder, which could confound the results for high BW (Dornhorst et al. 1992).
Table 2 gives approximate odds ratios and turning points for this association found in the four systematic reviews.
Table 2. Odds Ratios and Turning Points from Four Systematic Reviews
| Author of Review (in order of recency) | DM-Specific Outcomes? | Type of Association | Number of Studies Included | OR (BW<2,500g) | OR (BW>4,000g) | Turning Point (if continuous relationship) |
|---|---|---|---|---|---|---|
| Newsome et al. 2003 | Yes | Inverse | 48 | not calculated | not calculated | not calculated |
| Harder et al. 2007 | Yes | U-Shaped | 14 | 1.47 | 1.36 | Using smaller weight breakdowns, lowest risk was found for BW of between 3,500 and 4,000g. |
| Whincup et al. 2008 | Yes | Inverse | 30 (14 for meta-analysis) | 0.80 for every 1 kg increase | Of six subsets explored, 4 North American subpopulations showed a u-shaped relationship, with turning points between ~2,750 and 3,500g. | |
| Y. Song et al. 2012 | Yes | U-Shaped | 23 for meta-analysis | 0.78 for every 1 kg increase | Study did not conduct analysis to test for nonlinear relationship; rather it concluded from the evidence in other studies that the relationship between BW and DM risk is inverse and linear until 4,000g, at which point risk flattens and begins to increase again. | |
Except for in the studies on Native American populations, it appears the turning point for the relationship occurs at a relatively high BW. For example, looking at the latest available national BW surveys for two Southeast Asian countries, BW above 3,000g occurs rarely (Subramanyam, Ackerson, and Subramanian 2010; Bangladesh Bureau of Statistics and UNICEF 2005). This suggests that for LMICs, particularly those in Southeast Asia, there are too few babies being born at higher BW for the U-shaped relationship to be observed. Given the results, it is important to keep in mind the context of the population before assuming a relationship between BW and DM risk.
Potential Mediating Factors
Sex
When studies disaggregated analysis for child’s sex, the majority found no significant differences. A 2003 review of 48 studies that included risk disaggregated by sex found similar rates regardless of gender (Newsome et al. 2003). However, some evidence from other single studies showed that for certain populations, there may be greater variation. (Fall et al. 1995) found the relationship between BW and fasting glucose and insulin to be much stronger in women than men in England, while (Fall et al. 1998) found the opposite to be true in Mysore, India.
Gestational Age
Reflecting the current debate on what aspect of gestation drives the association with DM risk, several of the studies included controls for gestational age, fetal growth restriction, and birth length. For instance, (Phipps et al. 1993) found that the trends with BW were independent of gestational age, which was not found to be a significant predictor of risk. (Kaijser et al. 2009) confirmed an inverse association between BW and DM. The study also found an independent association between increased DM risk and preterm birth and fetal growth restriction. This suggests that if an infant is affected by both conditions, his or her risk would be multiplicative for DM.
Type, Rate, and Timing of Weight Gain
There also appears to be a significant effect on the relationship between LBW and DM risk in the presence of higher relative weight in later childhood, adolescence, or adulthood (Bavdekar et al. 1999; Raghupathy et al. 2010). Those born with LBW could potentially have a higher propensity of becoming overweight later in life; however, current evidence suggests that those born with high BW are at risk of becoming overweight, not those born with LBW (Baird et al. 2005). Studies in this area overwhelmingly come from developed countries, thus they cannot reflect the dynamics in the context of nutrition transition. Some evidence showed that regardless of total weight, when those born with LBW gain weight, it is more likely to be abdominal weight gain, a risk factor for metabolic imbalance (Schroeder, Martorell, and Flores 1999). Independence of effect was tested in several studies by adjusting for later-life BMI, which strengthened the association between LBW and DM (Newsome et al. 2003; Whincup et al. 2008).
In addition, analysis in a separate set of studies was extended to consider later-life weight gain on the association between BW and DM risk. These selected studies are described in Table 3. While not unanimous, the majority of these studies found an independent increased risk for developing DM among: a) those who had higher weight in later life (Adair et al. 2013; Crowther et al. 1998; Bonsdorff et al. 2013), and b) those who had more rapid weight gain in later life (Norris et al. 2012; Bhargava et al. 2004; Crowther et al. 1998). When combined, these results suggest that those most at risk of DM or pre-DM conditions are those born with LBW, who then gain weight at a rapid pace and attain a higher weight later in life relative to their peers.
Table 3. Selected Studies Finding Significant Modifying Effects of Weight Gain on BW-DM Risk Relationship
| Author – Date | Location | Condition | Modifier | Description |
|---|---|---|---|---|
| Adair et al. 2013 | Brazil, Guatemala, India, the Philippines, and South Africa | Fasting glucose, dysglycemia (IGT or DM) | Conditional weight & height (deviation from expected size based on own previous measures/child cohort - represents faster or slower relative weight gain) | Lower birthweight increased risk of dysglycemia (0·89 [0·81—0·98]), but linear growth and relative weight gain in childhood were not associated with dysglycemia. Adult conditional relative weight was also associated with dysglycemia (1.32 [1.20–1.45]). (ADD HEIGHT) |
| Sun et al. 2008 | USA | Metabolic syndrome | BMI | They found birthweight significantly and positively affects child BMI, which in turn positively affects adult obesity and risk of metabolic syndrome. The earliest age at which child weight diverged between the MS and non-MS group, independent of birthweight, was 8 years old for boys and 13 years old for girls. |
| Crowther et al. 1998 | South Africa | Fasting insulin, IR | BMI | Children born with LBW but who had high BMI at 7 years had higher insulin concentrations compared with those with LBW and low BMI at 7 years. There were also positive correlations between weight velocity and insulin resistance. |
| Bavdekar et al. 1999 | India | IR | Height/BMI | Taller height at 8 years predicted insulin resistance; the most insulin-resistant children were those who had short parents but had grown tall. The interaction of weight at eight years and birthweight was significant, with the LBW children who were now heaviest at higher risk for insulin resistance. |
| Bhargava et al. 2004 | India | IGT, DM | BMI & age at adiposity rebound | Those with IGT/DM typically had a low BMI up to two years of age, followed by an early adiposity rebound (the age after infancy when body mass starts to rise) and an accelerated increase in body-mass index until adulthood. |
| Fall et al. 2008 | India | IGT, DM, IR, Metabolic syndrome | BMI & age at adiposity rebound | Greater infant BMI/weight gain was associated with a lower risk of DM, especially in LBW infants, but it was also associated with an increased risk of metabolic syndrome and its components, a paradoxical finding that may be explained by the cohort's body composition during different age periods. Rapid BMI gain during childhood and adolescence was a risk factor for both disorders. |
| Norris et al. 2012 | Brazil, Guatemala, India, the Philippines, and South Africa | Fasting glucose/DM, IR | Conditional weight gain (deviation from expected size based on own previous measures/child cohort – represents faster or slower relative weight gain) | LBW and accelerated weight gain after 48 months are risk factors for adult glucose intolerance; greater CWG at 0–24 and 24–48 months and 48 months–adulthood predicted higher IR (all P , 0.001). Accelerated weight gain between 0 and 24 months did not predict glucose intolerance until adult waist circumference was controlled for, then it had an inverse association with fasting glucose/DM. |
| Bonsdorff et al. 2013 | Iceland | IR, DM | BMI | Compared with those with high birth weight and low BMI in midlife, the odds of diabetes was almost fivefold for individuals with low birth weight and high BMI (OR, 4.93; 95% CI, 2.14–11.37). Excessive weight gain in adulthood might be particularly detrimental to the health of old individuals with low birth weight. |
Although there is sufficient evidence to state that there is likely a compound risk of LBW, rapid weight gain, and high BMI in later life, it is unclear exactly when it is most risky for weight gain to occur during a person’s life. In the studies selected for this review, the age range for weight gain spans from one month to greater than 70 years. Findings were inconsistent for the risk in the late childhood–later adolescence period.
Some recent studies found no additional risk when weight gain occurred in the first two to four years of life (and in some cases, a very small protective effect for those born with LBW) but increased risk when additional weight gain occurred after that period (Norris et al. 2012; Bhargava et al. 2004; Fall et al. 2008). However, a separate literature review summarizing another set of evidence found DM risk beginning within the first two years of life, in some cases as early as the first six months (Y. Song et al. 2012).
Risk of DM and pre-DM conditions may also increase in the adolescent period (Bavdekar et al. 1999; Sun et al. 2008; Crowther et al. 1998). In a large study from Finland, the association with weight gain became significant in the seven- to 15-year-old period; the relationship to development of DM was strongest among those children born below 3,000g (Forsén et al. 2000). In yet another set of literature, the strongest, most significant effects occurred when weight gain or high BMI occurred in adulthood; however, one study found no effects prior to adulthood, while the other found a graduated effect over time (Adair et al. 2013; Fall et al. 2008).
Conclusions and Relevance
Because of low average BWs in LMICs, interventions to increase BW will likely decrease DM risk among these populations, with relatively little effect on risk at the highest end of BW. Focusing on the inverse relationship for BW below 4,000g, (Whincup et al. 2008) estimated that a population-wide intervention that increases BW as little as 100g – with larger increases (up to 200g) in populations with marginal nutrition – could translate into reductions in risk of DM by five to 10 percent. A change of ~100g in BW is not outside the realm of possibility via maternal supplementation, as has been documented by several studies; however, it appears that in Southeast Asia, the low starting weight of infants could reduce the absolute potential for change in BW (Gillespie and Haddad 2003; Abu-Saad and Fraser 2010; Christian 2003).
In other work conducted by SPRING, the three most effective interventions to reduce LBW/increase BW included maternal multiple micronutrient supplementation (MMS), balanced protein energy supplementation, and family planning to increase interpregnancy intervals (Pomeroy et al. 2014). The most effective of these appears to be MMS, which reduces risk of LBW by about 17 percent (when adjusted for the sociodemographic profile of that cohort). In that study, the authors were able to simulate the effects of such a reduction on cardiovascular disease risk later in life, using evidence of linkages throughout the life course.
In the case of DM, it is more difficult to make such estimations of reductions in mortality and morbidity. While it appears that the majority of the studies found a significant inverse relationship between BW (below a certain threshold) and DM risk , variance exists in the nature of the relationship, how persistent it is throughout the life cycle, and what modifies that risk in children and adults (Tinnion et al. 2013). Part of the reason this is a difficult relationship to understand is that DM has a multifactorial and dynamic etiology. This etiology includes strong contributions from childhood and adult body weight trajectory, lifestyle, and genetic factors (Whincup et al. 2008).
Beyond the greater heterogeneity in evidence on the initial effects of BW on DM, this review outlines the uncertainty on the compound DM risk of weight gain and/or high BMI on LBW. To further complicate the issue, this body of evidence also uses IR, dysglycemia, and other pre-DM conditions often in lieu of DM; however, as noted earlier, the pathway from these conditions to DM is not entirely clear.
To estimate risk reduction on adult DM-related mortality due to improved birth outcomes, one would require more concrete information on how modifiable this underlying risk is, by what factors, and when during the lifecycle it is most likely modified. It is also necessary to define clear progression from DM to mortality (preferably among several different demographic groups), as there are multiple routes available with uncertainty related to many of them (Fonseca 2009). Greater use of prospective cohort design rather than reliance on retrospective chart review would also improve estimates of DM risk and improve the discussion around causality.
This review highlights the variety of data on the effects of BW on DM and pre-DM condition risk in later life. While consensus is growing that BW does have a significant effect on DM risk, more work is needed to conclusively define that association in LMICs, in different ethnic groups, and across the spectrum of BW. In the meantime, all efforts to reduce LBW should continue, given its proven relationship with neonatal mortality (Yasmin et al. 2001; Ashworth 1998). The work in this review and elsewhere to improve estimates of the relationship between LBW and later life NCD risk should be considered as additional support for efforts to reduce LBW. The improved estimates will allow for more accurate accounting of the lives saved over the life course by improved birth outcomes.
Footnotes
1 This measure is a combined indicator of high glucose plus cholesterol, blood pressure, and waist circumference. It is used primarily to predict increased risk for heart disease and other health problems, such as diabetes and stroke.
References
Abu-Saad, Kathleen, and Drora Fraser. 2010. “Maternal Nutrition and Birth Outcomes.” Epidemiologic Reviews 32 (1): 5–25. doi:10.1093/epirev/mxq001.
Adair, Linda S, Caroline HD Fall, Clive Osmond, Aryeh D Stein, Reynaldo Martorell, Manuel Ramirez-Zea, Harshpal Singh Sachdev, et al. 2013. “Associations of Linear Growth and Relative Weight Gain during Early Life with Adult Health and Human Capital in Countries of Low and Middle Income: Findings from Five Birth Cohort Studies.” The Lancet 382 (9891): 525–34. doi:10.1016/S0140-6736(13)60103-8.
Anazawa, Sonoko, Yoshihito Atsumi, and Kempei Matsuoka. 2003. “Low Birth Weight and Development of Type 2 Diabetes in a Japanese Population.” Diabetes Care 26 (7): 2210–11.
Ashworth, A. 1998. “Effects of Intrauterine Growth Retardation on Mortality and Morbidity in Infants and Young Children.” European Journal of Clinical Nutrition 52 Suppl 1: S34–41; discussion S41–42.
Baird, Janis, David Fisher, Patricia Lucas, Jos Kleijnen, Helen Roberts, and Catherine Law. 2005. “Being Big or Growing Fast: Systematic Review of Size and Growth in Infancy and Later Obesity.” BMJ 331 (7522): 929. doi:10.1136/bmj.38586.411273.E0.
Bangladesh Bureau of Statistics, and UNICEF. 2005. National Low Birth Weight Survey of Bangladesh 2003-2004. Dhaka, Bangladesh: MSCWP/BBS/UNICEF.
Bavdekar, A, C S Yajnik, CH Fall, S Bapat, AN Pandit, V Deshpande, S Bhave, S D Kellingray, and C Joglekar. 1999. “Insulin Resistance Syndrome in 8-Year-Old Indian Children: Small at Birth, Big at 8 Years, or Both?” Diabetes 48 (12): 2422–29. doi:10.2337/diabetes.48.12.2422.
Bhargava, Santosh K., Harshpal Singh Sachdev, Caroline H.D. Fall, Clive Osmond, Ramakrishnan Lakshmy, David J.P. Barker, Sushant K. Dey Biswas, Siddharth Ramji, Dorairaj Prabhakaran, and Kolli Srinath Reddy. 2004. “Relation of Serial Changes in Childhood Body-Mass Index to Impaired Glucose Tolerance in Young Adulthood.” N Engl J Med 350 (9): 865–75. doi:10.1056/NEJMoa035698.
Boney, Charlotte M, Anila Verma, Richard Tucker, and Betty R Vohr. 2005. “Metabolic Syndrome in Childhood: Association with Birth Weight, Maternal Obesity, and Gestational Diabetes Mellitus.” Pediatrics 115 (3): e290–96. doi:10.1542/peds.2004-1808.
Bonsdorff, Mikaela B, Majon Muller, Thor Aspelund, Melissa Garcia, Gudny Eiriksdottir, Taina Rantanen, Ingibjörg Gunnarsdottir, et al. 2013. “Persistence of the Effect of Birth Size on Dysglycaemia and Type 2 Diabetes in Old Age: AGES-Reykjavik Study.” AGE 35 (4): 1401–9. doi:10.1007/s11357-012-9427-5.
Carpenter, Marshall W. 2007. “Gestational Diabetes, Pregnancy Hypertension, and Late Vascular Disease.” Diabetes Care 30 Suppl 2 (July): S246–50. doi:10.2337/dc07-s224.
Choi, CS, C-H Kim, WJ Lee, JY Park, SK Hong, MG Lee, SW Park, and K-U. Lee. 2000. “Association between Birth Weight and Insulin Sensitivity in Healthy Young Men in Korea: Role of Visceral Adiposity.” Diabetes Research and Clinical Practice 49 (1): 53–59. doi:10.1016/S0168-8227(00)00131-5.
Chowdhury, Tahseen A, and Graham A Hitman. 2007. “Type 2 Diabetes in People of South Asian Origin: Potential Strategies for Prevention.” British Journal of Diabetes and Vascular Disease 7 (6): 279–82.
Christian, Parul. 2003. “Effects of Alternative Maternal Micronutrient Supplements on Low Birth Weight in Rural Nepal: Double Blind Randomised Community Trial.” BMJ 326 (7389): 571–571. doi:10.1136/bmj.326.7389.571.
Crowther, N J, N Cameron, J Trusler, and I P Gray. 1998. “Association between Poor Glucose Tolerance and Rapid Post Natal Weight Gain in Seven-Year-Old Children.” Diabetologia 41 (10): 1163–67. doi:10.1007/s001250051046.
Curioni, C C, and P M Lourenço. 2005. “Long-Term Weight Loss after Diet and Exercise: A Systematic Review.” International Journal of Obesity 29 (10): 1168–74. doi:10.1038/sj.ijo.0803015.
Darendeliler, Feyza, Firdevs Bas, Ruveyde Bundak, Asuman Coban, Ozlem Sancakli, Sema Kabatas Eryilmaz, Banu Kucukemre, et al. 2008. “Insulin Resistance and Body Composition in Preterm Born Children during Prepubertal Ages.” Clinical Endocrinology 68 (5): 773–79. doi:10.1111/j.1365-2265.2007.03119.x.
Deng, Hong-Zhu, Yan-Hong Li, Zhe Su, Hua-Mei Ma, Yue-Fang Huang, Hong-Shan Chen, and Min-Lian Du. 2011. “Association between Height and Weight Catch-up Growth with Insulin Resistance in Pre-Pubertal Chinese Children Born Small for Gestational Age at Two Different Ages.” European Journal of Pediatrics 170 (1): 75–80. doi:10.1007/s00431-010-1274-8.
Dornhorst, A, C M Paterson, S Nicholls, J Wadsworth, D C Chiu, R S Elkeles, D G Johnston, and R W Beard. 1992. “High Prevalence of Gestational Diabetes in Women from Ethnic Minority Groups.” Diabetic Medicine: A Journal of the British Diabetic Association 9 (9): 820–25.
Edelman, Steven. 2008. “GLYCEMIC CONTROL: THE FUTURE OF DIABETES MANAGEMENT.” Powerpoint presented at the Medscape Multispecialty Education Series, Online. www.medscape.org/viewarticle/412864.
Eriksson, J G, T Forsén, J Tuomilehto, C Osmond, and D J P Barker. 2003. “Early Adiposity Rebound in Childhood and Risk of Type 2 Diabetes in Adult Life.” Diabetologia 46 (2): 190–94. doi:10.1007/s00125-002-1012-5.
Fall, Caroline H D, Harshpal Singh Sachdev, Clive Osmond, Ramakrishnan Lakshmy, Sushant Dey Biswas, Dorairaj Prabhakaran, Nikhil Tandon, et al. 2008. “Adult Metabolic Syndrome and Impaired Glucose Tolerance Are Associated With Different Patterns of BMI Gain During Infancy Data from the New Delhi Birth Cohort.” Diabetes Care 31 (12): 2349–56. doi:10.2337/dc08-0911.
Fall, C H D, C Osmond, D J P Barker, P M S Clark, C N Hales, Y Stirling, and T W Meade. 1995. “Fetal and Infant Growth and Cardiovascular Risk Factors in Women.” BMJ 310 (6977): 428–32. doi:10.1136/bmj.310.6977.428.
Fall, CHD., CE. Stein, K Kumaran, V Cox, C Osmond, D J P Barker, and C N Hales. 1998. “Size at Birth, Maternal Weight, and Type 2 Diabetes in South India.” Diabetic Medicine 15 (3): 220–27. doi:10.1002/(SICI)1096-9136(199803)15:3<220::AID-DIA544>3.0.CO;2-O.
Finken, M . J, M G Keijzer-Veen, W Dekker, M Frölich, E T M Hille, J A Romijn, and J M Wit. 2006. “Preterm Birth and Later Insulin Resistance: Effects of Birth Weight and Postnatal Growth in a Population Based Longitudinal Study from Birth into Adult Life.” Diabetologia 49 (3): 478–85. doi:10.1007/s00125-005-0118-y.
Fonseca, VA. 2009. “Defining and Characterizing the Progression of Type 2 Diabetes.” Diabetes Care 32 (suppl_2): S151–56. doi:10.2337/dc09-S301.
Forsén, T, J. Eriksson, J Tuomilehto, A Reunanen, C Osmond, and D Barker. 2000. “The Fetal and Childhood Growth of Persons Who Develop Type 2 Diabetes.” Annals of Internal Medicine 133 (3): 176–82.
Freedman, D S, L K Khan, W H Dietz, S R Srinivasan, and G S Berenson. 2001. “Relationship of Childhood Obesity to Coronary Heart Disease Risk Factors in Adulthood: The Bogalusa Heart Study.” Pediatrics 108 (3): 712–18.
Gillespie, Stuart, and Lawrence Haddad. 2003. The Double Burden of Malnutrition in Asia: Causes, Consequences, and Solutions. SAGE Publications, India.
Godfrey, Keith M, Peter D Gluckman, and Mark A Hanson. 2010. “Developmental Origins of Metabolic Disease: Life Course and Intergenerational Perspectives.” Trends in Endocrinology & Metabolism 21 (4): 199–205. doi:10.1016/j.tem.2009.12.008.
Hamid Zargar, Abdul, Arshad Iqbal Wani, Shariq Rashid Masoodi, Bashir Ahmad Laway, and Mir Iftikhar Bashir. 1999. “Mortality in Diabetes Mellitus—data from a Developing Region of the World.” Diabetes Research and Clinical Practice 43 (1): 67–74. doi:10.1016/S0168-8227(98)00112-0.
Harder, T, E Rodekamp, K Schellong, J W Dudenhausen, and A Plagemann. 2007. “Birth Weight and Subsequent Risk of Type 2 Diabetes: A Meta-Analysis.” American Journal of Epidemiology 165 (8): 849–57. doi:10.1093/aje/kwk071.
Hattersley, A T, and J E Tooke. 1999. “The Fetal Insulin Hypothesis: An Alternative Explanation of the Association of Low Birthweight with Diabetes and Vascular Disease.” Lancet 353 (9166): 1789–92. doi:10.1016/S0140-6736(98)07546-1.
Holman, Rury R, Harald Sourij, and Robert M Califf. 2014. “Cardiovascular Outcome Trials of Glucose-Lowering Drugs or Strategies in Type 2 Diabetes.” The Lancet 383 (9933): 2008–17. doi:10.1016/S0140-6736(14)60794-7.
Ho, Mandy, Sarah P Garnett, Louise A Baur, Tracy Burrows, Laura Stewart, Melinda Neve, and Clare Collins. 2013. “Impact of Dietary and Exercise Interventions on Weight Change and Metabolic Outcomes in Obese Children and Adolescents: A Systematic Review and Meta-Analysis of Randomized Trials.” JAMA Pediatrics 167 (8): 759. doi:10.1001/jamapediatrics.2013.1453.
Kaijser, Magnus, Anna-Karin Edstedt Bonamy, Olof Akre, Sven Cnattingius, Fredrik Granath, Mikael Norman, and Anders Ekbom. 2009. “Perinatal Risk Factors for Diabetes in Later Life.” Diabetes 58 (3): 523–26. doi:10.2337/db08-0558.
Karaolis-Danckert, Nadina, Anette E Buyken, Katja Bolzenius, Carolina Perim De Faria, Michael J Lentze, and Anja Kroke. 2006. “Rapid Growth Among Term Children Whose Birth Weight Was Appropriate for Gestational Age Has a Longer Lasting Effect on Body Fat Percentage Than on Body Mass Index.” The American Journal of Clinical Nutrition 84 (6): 1449–55.
Kim, Chul-Hee, Hong-Kyu Kim, Sung-Jin Bae, Eun-Hee Kim, and Joong-Yeol Park. 2012. “Independent Impact of Body Mass Index and Metabolic Syndrome on the Risk of Type 2 Diabetes in Koreans.” Metabolic Syndrome and Related Disorders 10 (5): 321–25. doi:10.1089/met.2011.0143.
Kuzawa, Christopher W, Pedro C Hallal, Linda Adair, Santosh K Bhargava, Caroline H D Fall, Nanette Lee, Shane A Norris, et al. 2012. “Birth Weight, Postnatal Weight Gain, and Adult Body Composition in Five Low and Middle Income Countries.” American Journal of Human Biology 24 (1): 5–13. doi:10.1002/ajhb.21227.
Ley, Sylvia H, Osama Hamdy, Viswanathan Mohan, and Frank B Hu. 2014. “Prevention and Management of Type 2 Diabetes: Dietary Components and Nutritional Strategies.” The Lancet 383 (9933): 1999–2007. doi:10.1016/S0140-6736(14)60613-9.
Li, R, P. Zhang, L E Barker, F M Chowdhury, and X Zhang. 2010. “Cost-Effectiveness of Interventions to Prevent and Control Diabetes Mellitus: A Systematic Review.” Diabetes Care 33 (8): 1872–94. doi:10.2337/dc10-0843.
Monasta, L, G. D Batty, A Macaluso, L Ronfani, V Lutje, A Bavcar, F J van Lenthe, J Brug, and A Cattaneo. 2011. “Interventions for the Prevention of Overweight and Obesity in Preschool Children: A Systematic Review of Randomized Controlled Trials: Prevention of Obesity in Preschool Children.” Obesity Reviews 12 (5): e107–18. doi:10.1111/j.1467-789X.2010.00774.x.
Newsome, C A, A W Shiell, C H D Fall, D I W Phillips, R Shier, and C M Law. 2003. “Is Birth Weight Related to Later Glucose and Insulin Metabolism?—a Systematic Review.” Diabetic Medicine 20 (5): 339–48. doi:10.1046/j.1464-5491.2003.00871.x.
Norris, Shane A, Clive Osmond, Denise Gigante, Christopher W Kuzawa, Lakshmy Ramakrishnan, Nanette R Lee, Manual Ramirez-Zea, et al. 2012. “Size at Birth, Weight Gain in Infancy and Childhood, and Adult Diabetes Risk in Five Low- or Middle-Income Country Birth Cohorts.” Diabetes Care 35 (1): 72–79. doi:10.2337/dc11-0456.
Parkinson, James R C, Matthew J Hyde, Chris Gale, Shalini Santhakumaran, and Neena Modi. 2013. “Preterm Birth and the Metabolic Syndrome in Adult Life: A Systematic Review and Meta-Analysis.” Pediatrics 131 (4): e1240–63.
Perneger, T V, F L Brancati, P K Whelton, and M J Klag. 1994. “End-Stage Renal Disease Attributable to Diabetes Mellitus.” Annals of Internal Medicine 121 (12): 912–18.
Peters, Sanne A E, Rachel R Huxley, and Mark Woodward. 2014. “Diabetes as a Risk Factor for Stroke in Women Compared with Men: A Systematic Review and Meta-Analysis of 64 Cohorts, Including 775 385 Individuals and 12 539 Strokes.” The Lancet 383 (9933): 1973–80. doi:10.1016/S0140-6736(14)60040-4.
Phipps, K, D. Barker, C Hales, C Fall, C Osmond, and P Clark. 1993. “Fetal Growth and Impaired Glucose Tolerance in Men and Women.” Diabetologia 36 (3): 225–28. doi:10.1007/BF00399954.
Pomeroy, Amanda, Marc Cunningham, Alexis D’Agostino, Warren Stevens, and Jolene Wun. 2014. Phase I Report of the Early-Life Nutrition Linkages to Noncommunicable Disease (ELN-NCD) Model: Maternal Interventions to Improve Birthweight and Gestational Age, Bangladesh. Arlington, VA: SPRING Project. http://www.spring-nutrition.org/sites/default/files/publications/reports....
Rademacher, Erin R, David R Jacobs, Antoinette Moran, Julia Steinberger, Ronald J Prineas, and Alan Sinaiko. 2009. “Relation of Blood Pressure and Body Mass Index during Childhood to Cardiovascular Risk Factor Levels in Young Adults.” Journal of Hypertension 27 (9): 1766–74. doi:10.1097/HJH.0b013e32832e8cfa.
Raghupathy, Palany, Belavendra Antonisamy, Finney S Geethanjali, Julia Saperia, Samantha D Leary, G. Priya, Joseph Richard, David JP Barker, and Caroline HD Fall. 2010. “Glucose Tolerance, Insulin Resistance and Insulin Secretion in Young South Indian Adults: Relationships to Parental Size, Neonatal Size and Childhood Body Mass Index.” Diabetes Research and Clinical Practice 87 (2): 283–92. doi:10.1016/j.diabres.2009.11.015.
Rotteveel, J., M. M. van Weissenbruch, J. W. R. Twisk, and H. A. Delemarre-Van de Waal. 2008. “Infant and Childhood Growth Patterns, Insulin Sensitivity, and Blood Pressure in Prematurely Born Young Adults.” PEDIATRICS 122 (2): 313–21. doi:10.1542/peds.2007-2012.
Sachdev, Harshpal S., Caroline HD Fall, Clive Osmond, Ramakrishnan Lakshmy, Sushant K. Dey Biswas, Samantha D. Leary, Kolli Srinath Reddy, David JP Barker, and Santosh K. Bhargava. 2005. “Anthropometric Indicators of Body Composition in Young Adults: Relation to Size at Birth and Serial Measurements of Body Mass Index in Childhood in the New Delhi Birth Cohort.” The American Journal of Clinical Nutrition 82 (2): 456–66.Schroeder, D. G., R. Martorell, and R. Flores. 1999. “Infant and Child Growth and Fatness and Fat Distribution in Guatemalan Adults.” American Journal of Epidemiology 149 (2): 177–85.
Schroeder, D G, R Martorell, and R Flores. 1999. “Infant and Child Growth and Fatness and Fat Distribution in Guatemalan Adults.” American Journal of Epidemiology 149 (2): 177–85.
Shaw, J.E., R.A. Sicree, and P.Z. Zimmet. 2010. “Global Estimates of the Prevalence of Diabetes for 2010 and 2030.” Diabetes Research and Clinical Practice 87 (1): 4–14. doi:10.1016/j.diabres.2009.10.007.
Shi, Yuankai, and Frank B Hu. 2014. “The Global Implications of Diabetes and Cancer.” The Lancet 383 (9933): 1947–48. doi:10.1016/S0140-6736(14)60886-2.
Song, Soon H, and Colin A Hardisty. 2008. “Early-Onset Type 2 Diabetes Mellitus: An Increasing Phenomenon of Elevated Cardiovascular Risk.” Expert Review of Cardiovascular Therapy 6 (3): 315–22. doi:10.1586/14779072.6.3.315.
Song, Y, L Wang, E Yeung, and Cuilin Zhang. 2012. “Body Size at Birth and Risk of Type 2 Diabetes in Adult Life.” In Handbook of Anthropometry, pp20732090. New York, NY: Springer New York.
Stern, Michael P, Ken Williams, Clicerio González-Villalpando, Kelly J Hunt, and Steven M Haffner. 2004. “Does the Metabolic Syndrome Improve Identification of Individuals at Risk of Type 2 Diabetes And/or Cardiovascular Disease?” Diabetes Care 27 (11): 2676–81. doi:10.2337/diacare.27.11.2676.
Subramanyam, Malavika A, Leland K Ackerson, and S V Subramanian. 2010. “Patterning in Birthweight in India: Analysis of Maternal Recall and Health Card Data.” PLoS ONE 5 (7): e11424. doi:10.1371/journal.pone.0011424.
Sugihara, Shigetaka, Nozomu Sasaki, Shin Amemiya, Hitoshi Kohno, Toshiaki Tanaka, and Nobuo Matsuura. 2008. “Analysis of Weight at Birth and at Diagnosis of Childhood-Onset Type 2 Diabetes Mellitus in Japan.” Pediatric Diabetes 9 (4 Pt 1): 285–90. doi:10.1111/j.1399-5448.2008.00402.x.
Sung, Ki-Chul, Woo-Shin Jeong, Sarah H Wild, and Christopher D Byrne. 2012. “Combined Influence of Insulin Resistance, Overweight/obesity, and Fatty Liver as Risk Factors for Type 2 Diabetes.” Diabetes Care 35 (4): 717–22. doi:10.2337/dc11-1853.
Sun, Shumei S, Ruohong Liang, Terry T-K Huang, Stephen R Daniels, Silva Arslanian, Kiang Liu, Gilman D Grave, and Roger M Siervogel. 2008. “Childhood Obesity Predicts Adult Metabolic Syndrome: The Fels Longitudinal Study.” The Journal of Pediatrics 152 (2): 191–200.e1. doi:10.1016/j.jpeds.2007.07.055.
The Lancet. 2014. “Back to Basics for Diabetes.” The Lancet 383 (9933): 1945. doi:10.1016/S0140-6736(14)60937-5.
Timæus, Ian M. 2012. “Stunting and Obesity in Childhood: A Reassessment Using Longitudinal Data from South Africa.” International Journal of Epidemiology 41 (3): 764–72. doi:10.1093/ije/dys026.
Tinnion, Robert, Jenna Gillone, Timothy Cheetham, and Nicholas Embleton. 2013. “Preterm Birth and Subsequent Insulin Sensitivity: A Systematic Review.” Archives of Disease in Childhood, December, archdischild – 2013–304615. doi:10.1136/archdischild-2013-304615.
Wei, Jung-Nan, Fung-Chang Sung, Chung-Yi Li, Chia-Hsuin Chang, Ruey-Shiung Lin, Chau-Ching Lin, Chuan-Chi Chiang, and Lee-Ming Chuang. 2003. “Low Birth Weight and High Birth Weight Infants Are Both at an Increased Risk to Have Type 2 Diabetes among Schoolchildren in Taiwan.” Diabetes Care 26 (2): 343–48.
Whincup, P H, S J Kaye, C G Owen, R Huxley, D G Cook, S Anazawa, E. Barrett-Connor, et al. 2008. “Birth Weight and Risk of Type 2 Diabetes: A Systematic Review.” JAMA: The Journal of the American Medical Association 300 (24): 2886–97. doi:10.1001/jama.2008.886.
Yajnik, C S. 2002. “The Lifecycle Effects of Nutrition and Body Size on Adult Adiposity, Diabetes and Cardiovascular Disease.” Obesity Reviews 3 (3): 217–24. doi:10.1046/j.1467-789X.2002.00072.x.
Yasmin, S, D Osrin, E Paul, and A Costello. 2001. “Neonatal Mortality of Low-Birth-Weight Infants in Bangladesh.” Bulletin of the World Health Organization 79 (7): 608–14.
Yoon, Kun-Ho, Jin-Hee Lee, Ji-Won Kim, Jae Hyoung Cho, Yoon-Hee Choi, Seung-Hyun Ko, Paul Zimmet, and Ho-Young Son. 2006. “Epidemic Obesity and Type 2 Diabetes in Asia.” The Lancet 368 (9548): 1681–88. doi:10.1016/S0140-6736(06)69703-1.
Zargar, Abdul Hamid, Arshad Iqbal Wani, Shariq Rashid Masoodi, Bashir Ahmad Laway, and Mir Iftikhar Bashir. 1999. “Mortality in Diabetes Mellitus—Data from a Developing Region of the World.” Diabetes Research and Clinical Practice 43 (1): 67–74. doi:10.1016/S0168-8227(98)00112-0.