Findings from the Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project
Summary

Anemia is a serious public health problem caused by multiple factors. Understanding what causes anemia in a particular context is critical to designing effective anemia programs and monitoring their impact.
Anemia is a condition characterized by insufficient hemoglobin, a protein responsible for transporting oxygen in the blood. Anemia can be caused by infection, inflammation, micronutrient deficiencies, and genetics. Therefore, a data-driven and multi-sectoral approach is required to design and implement programs that target the specific causes of anemia within a given context. However, the ability to accurately assess the factors contributing to anemia can make such a comprehensive approach difficult.
In particular, estimating iron and vitamin A deficiencies— two of the more common nutritional deficiencies that can cause anemia—is difficult in settings where there is a high burden of infection. This is because inflammation, caused by infections and chronic conditions, can modify the biomarker values used to assess iron and vitamin A deficiencies.
The Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) project set out to improve the interpretation of iron and vitamin A biomarker results and our understanding of the main risk factors for anemia. The BRINDA project recently published findings that have significant implications for anemia programming, especially programs aimed at addressing micronutrient deficiencies. The findings indicate the importance of applying adjustments for inflammation to iron and vitamin A biomarkers to correctly interpret the prevalence of these deficiencies and their association with anemia. The BRINDA project also identified key risk factors for anemia across several countries. In this brief, we summarize these findings and explain their relevance for anemia programming.
The brief is intended for programmers and policymakers involved in—
- planning a survey that measures anemia or micronutrient deficiencies
- interpreting results of surveys that measure anemia or micronutrient deficiencies (e.g., conducting a landscape analysis)
- designing and implementing programs to reduce anemia and micronutrient deficiencies.
Key Findings from the BRINDA Project
- Surveys that only measure anemia do not provide sufficient information about the factors causing anemia, nor can they provide information on the nutritional situation in a country. Data on the prevalence of anemia are frequently wrongly interpreted to indicate iron deficiency.
- There is a need for more comprehensive assessment of the causes of anemia to be able to design appropriate interventions.
- Surveys should measure a range of risk factors depending on the setting, such as micronutrient deficiencies, inflammation, infectious diseases, and genetics, to understand what is driving anemia.
- Risk factors for anemia vary by setting, although iron deficiency remains a major contributing factor for anemia in many countries.
- Accurate estimates of iron and vitamin A deficiencies are necessary to design interventions that address the nutritional causes of anemia in a country. A well-designed program will have limited impact if it does not address an underlying need.
- Estimating iron and vitamin A deficiency without adjusting for inflammation leads to inaccurate estimates. Adjustments to iron and vitamin A biomarkers must be applied to interpret the data. Future research will determine if adjustments are needed for other micronutrient biomarkers.
Background
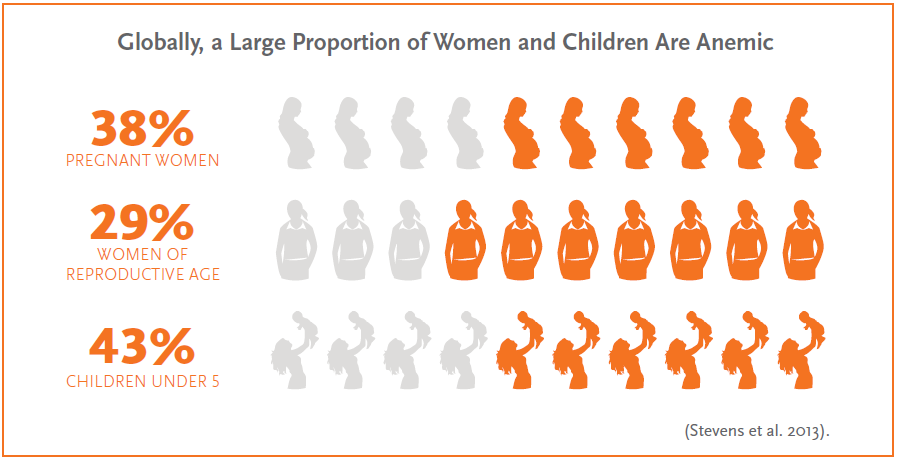
Anemia is one of the leading causes of disability worldwide.
Anemia is one of the most common public health problems globally and very few countries are making sufficient progress to meet the anemia reduction targets set by the World Health Assembly.1, 2 Anemia can result in low birthweight and premature birth, decreased productivity and work capacity, and increased risk of maternal and child mortality.3, 4
Many factors can cause anemia, individually or in combination.
The main causes of anemia are micronutrient deficiencies, infections, inflammation, and genetic disorders.5 A deficiency in the micronutrient iron, which is essential for the production of hemoglobin, is a common cause of anemia. Other micronutrient deficiencies, such as vitamin A, zinc, folate, and B12, may also contribute to anemia, but the extent to which they do so is unknown. Infections, such as malaria, can also cause anemia by destroying red blood cells or decreasing their production, while other infections can cause blood loss (e.g., hookworm). Inflammation, which occurs during acute infections and chronic conditions, can also cause anemia by altering the absorption and utilization of micronutrients, and by decreasing red blood cell production. Lastly, genetic blood disorders (e.g., thalassemia, sickle cell)—which create abnormalities in the function, structure, or production of red blood cells—can also result in anemia.
KEY TERMS
Acute phase proteins: proteins produced by the liver as part of the immune response to protect and repair damage in the body. They are classified as positive or negative depending on whether they rise or fall, respectively, during an immune response.6
AGP: acute phase protein that rises slowly and remains elevated for a prolonged period in response to inflammatory stimulus. It can be assessed in blood to measure inflammation.6
Anemia: a condition characterized by low values of hemoglobin in red blood cells.7
Biomarker: a measurable indicator of a biological state or condition.8
CRP: acute phase protein that rises and falls quickly in response to inflammatory stimulus. It can be assessed in blood to measure inflammation.9
Ferritin: a protein that stores iron. It can be assessed in blood and used to measure iron status. It is also a positive acute phase protein.10
Hemoglobin: a protein in red blood cells that is necessary for carrying oxygen in the body. It can be assessed in blood to measure anemia.7
Inflammation: the body's response to infection or tissue injury. It may be acute (self-limiting and resolves rapidly) or chronic (body fails to regulate inflammatory process and becomes persistent).8
Inflammatory response: a process triggered by the body in response to stressors, including microbial invasion, tissue injury, and disease.8
Retinol: predominant circulating form of vitamin A and attached to retinol-binding protein in blood. It is used to assess vitamin A status.11
Retinol-binding protein: a carrier protein for retinol that is highly correlated with retinol and is used to assess vitamin A status. It is also a negative acute phase protein.11
Hemoglobin tells us if anemia exists, but not what is causing it.
Anemia is most commonly measured by assessing hemoglobin levels in the blood, but this biomarker does not provide information on what is causing the anemia. Measurement of micronutrient biomarkers (e.g., for iron, vitamin A, folate in serum or plasma), usually through a blood test, can identify the presence of micronutrient deficiencies. Infections can be assessed through rapid diagnostic kits (e.g., malaria, HIV), examination of stool or urine samples (e.g., helminth infections), or by reported illness. Inflammation can be assessed by measuring the levels of acute phase proteins found during periods of inflammation in the blood, such as C-reactive protein (CRP) and alpha-1 acid glycoprotein (AGP). The presence of genetic blood disorders can be assessed using molecular techniques. An assessment of all these potential causes of anemia, or risk factors, can help us better understand the important drivers of anemia and the extent to which individual risk factors may be a problem.
Iron deficiency has generally been assumed to be the main cause of anemia, but we are learning that this may not be true in all contexts.
Historically, iron deficiency was assumed to cause about 50 percent of anemia. Consequently, anemia prevention and control programs often rely primarily on reducing iron deficiency through the provision of iron supplementation. More recently, as assessment of the anemia situation in countries has improved, we are learning that many factors play a role and that we need to identify the multiple causes in each setting. Better identification of the key drivers of anemia, including more accurate estimates of the extent to which micronutrient deficiencies contribute to anemia, is needed to design effective interventions and monitor impact.
Micronutrient deficiencies can be assessed using biomarkers in the blood.
Assessing micronutrient deficiencies is particularly challenging. This brief summarizes why that is and provides approaches for assessing two such micronutrients: iron and vitamin A. The World Health Organization (WHO) recommends using ferritin levels in the blood as the primary measure of population-level iron deficiency, complemented by assessment of hemoglobin levels in the blood.10 The WHO recommends using retinol levels in the blood to assess population vitamin A status,11 although retinol-binding protein (RBP) assessment in the blood is also increasingly being used as a proxy for retinol, as one molecule of RBP usually carries one molecule of retinol, and it is very rare to have free retinol in circulation.11 As noted previously, there are other micronutrients found to be associated with anemia, but assessment of these biomarkers is beyond the scope of this brief.
Using acute phase proteins as biomarkers for iron and vitamin A can be misleading in cases where there is inflammation.
Iron (ferritin) and vitamin A biomarkers (RBP) are difficult to interpret in settings with a high burden of infections because the former is a positive acute phase protein (increases in the presence of inflammation) and the latter is a negative acute phase protein (decreases in the presence of inflammation). When the body is in an inflammatory state, the blood levels of ferritin increase whereas the levels of retinol and RBP decease as part of the immune response, irrespective of micronutrient status.6 Under an inflammatory state, ferritin, retinol (which is attached to RBP), and RBP no longer accurately measure iron and vitamin A status; therefore, mathematical adjustments are necessary. Adjustments can be made to ferritin, retinol, and RBP levels by measuring other acute phase proteins not related to iron or vitamin A status. Two such acute phase proteins are CRP and AGP. CRP is thought to rise quickly in response to an infection but only remains elevated for a short period, whereas AGP is thought to rise slowly in response to an infection and remain elevated longer. Other micronutrient biomarkers may also be influenced by inflammation, but an examination of those is beyond the scope of this brief.
Methods Used to Inform Findings
About the BRINDA project
The BRINDA project was formed in 2012 to improve the design and monitoring of anemia-related interventions by—
- characterizing the relationship of inflammation with iron and vitamin A biomarker values
- identifying approaches to account for the effects of inflammation on iron and vitamin A biomarker values
- obtaining reliable estimates of the nutritional and non-nutritional risk factors of anemia.
The first phase of the BRINDA project was led by the Centers for Disease Control and Prevention (CDC), Global Alliance for Improved Nutrition (GAIN), and National Institute of Child Health and Human Development (NICHD), with representation by individuals from countries that contributed data.
Data sets included
BRINDA project researchers identified nationally and regionally representative nutrition surveys from the last 10 years that included preschool children (6–59 months), non-pregnant women of childbearing age (15–49 years), or both. To be included in the study, these surveys had to have data on the following biomarkers:
- anemia (measured by hemoglobin)
- iron (measured by ferritin or soluble transferrin receptor) or vitamin A (measured by RBP or retinol)
- inflammation (measured by AGP, CRP, or both).
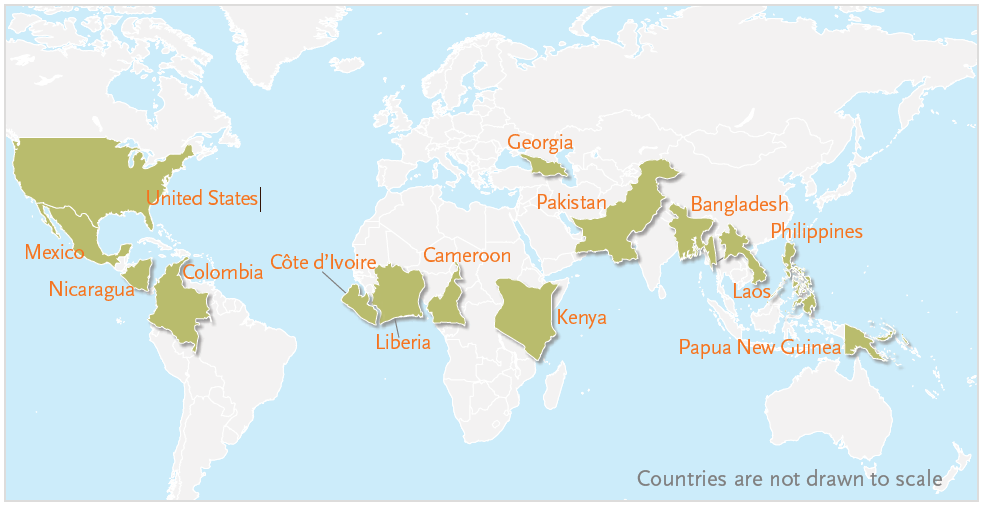
The BRINDA project used a total of 16 data sets from 14 countries (n=29,766 children and 25,731 women) that met the inclusion criteria (figure 1). Among the 16 datasets, 12 were national surveys while 4 were sub-national surveys. All 16 surveys provided data for children and 10 provided data for women. The BRINDA project data sets are not globally representative, but do represent a large range of national and sub-national information from all six WHO geographic regions.
Methods for adjusting estimates of iron and vitamin A deficiency
To improve estimates of iron (ferritin)a and vitamin A deficiency (RBP), the researchers compared two statistically grounded approaches to adjust ferritin and RBP values for inflammation (AGP and CRP). The first approach, "correction factor," adjusts the ferritin and RBP values for all individuals whose AGP and CRP values pass a clinically defined threshold (AGP >1 g/L and CRP > 5mg/L).12, 13 This approach does not take into account the relationship between ferritin and RBP with AGP and CRP values at levels below the clinically defined thresholds for AGP and CRP.
The second approach, "regression correction," adjusts the ferritin and RBP values along a continuum, which captures the relationship of these nutrient biomarkers with AGP or CRP values at both low and high levels of AGP or CRP values. This approach makes adjustments to ferritin and RBP values at levels below the clinically defined thresholds for AGP and CRP.b
Figure 1. The BRINDA Project Dataset Included Countries from Every Region in the World
Methods for assessing risk factors for anemia
Researchers identified primary risk factors for anemia using bivariate and multivariable logistic regressions. Risk factors included in the model were demographics, nutritional deficiencies, inflammation, and infections. Regression correction adjustments were made to the iron (ferritin) and vitamin A biomarkers (either RBP or retinol depending on the survey) for inflammation prior to including these risk factors in the model.
The risk factors associated with anemia were determined for each individual country included in the BRINDA database. The researchers also pooled the data by categorizing the countries from the BRINDA database into groups based on their risk of infectious disease burden (low, medium, high, and very high for children and low, medium, and high for women).This categorization was determined by assigning scores to the countries based on their prevalence of malaria, diarrhea, schistosomiasis, and on the availability of sanitation and safe drinking water, adapted from Petry et al.14
BRINDA Project Articles
Engle-Stone, Reina, Grant J. Aaron, Jin Huang, James P. Wirth, Sorrel M.L. Namaste, Anne M. Williams, Janet M. Peerson, et al. 2017. "Predictors of Anemia in Preschool Children: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142323. doi:10.3945/ajcn.116.142323.
Larson, Leila M., Sorrel M.L. Namaste, Anne M. Williams, Reina Engle-Stone, O. Yaw Addo, Parminder S. Suchdev, James P. Wirth, Victor Temple, Mary Serdula, and Christine A. Northrop-Clewes. 2017. "Adjusting Retinol-Binding Protein Concentrations for Inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142166. doi:10.3945/ajcn.116.142166.
Mei, Zuguo, Sorrel M.L. Namaste, Mary Serdula, Parminder S. Suchdev, Fabian Rohner, Rafael Flores-Ayala, O. Yaw Addo, and Daniel J. Raiten. 2017. "Adjusting Total Body Iron for Inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142307. doi:10.3945/ajcn.116.142307.
Merrill, Rebecca D., Rachel M. Burke, Christine A. Northrop-Clewes, Pura Rayco-Solon, Rafael Flores-Ayala, Sorrel M.L. Namaste, Mary K. Serdula, and Parminder S. Suchdev. 2017. "Factors Associated with Inflammation in Preschool Children and Women of Reproductive Age: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142315. doi:10.3945/ajcn.116.142315.
Namaste, Sorrel M.L., Grant J. Aaron, Ravi Varadhan, Janet M. Peerson, and Parminder S. Suchdev. 2017. "Methodologic Approach for the Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142273. doi:10.3945/ajcn.116.142273.
Namaste, Sorrel M.L., Fabian Rohner, Jin Huang, Nivedita L. Bhushan, Rafael Flores-Ayala, Roland Kupka, Zuguo Mei, et al. 2017. "Adjusting Ferritin Concentrations for Inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn141762. doi:10.3945/ajcn.116.141762.
Rohner, Fabian, Sorrel M.L. Namaste, Leila M. Larson, O. Yaw Addo, Zuguo Mei, Parminder S. Suchdev, Anne M. Williams, et al. 2017. "Adjusting Soluble Transferrin Receptor Concentrations for Inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142232. doi:10.3945/ajcn.116.142232.
Stoltzfus, Rebecca J. and Rolf Klemm. 2017. "Research, Policy, and Programmatic Considerations from the Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn142372. doi:10.3945/ajcn.116.142372.
Suchdev, Parminder S., Sorrel M.L. Namaste, Grant J. Aaron, Daniel J. Raiten, Kenneth H. Brown, Rafael Flores-Ayala, and on behalf of the BRINDA Working Group. 2016. "Overview of the Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." Advances in Nutrition: An International Review Journal 7 (2): 349–56. doi:10.3945/an.115.010215.
Wirth, James P., Bradley A. Woodruff, Reina Engle-Stone, Sorrel M.L. Namaste, Victor J. Temple, Nicolai Petry, Barbara Macdonald, Parminder S. Suchdev, Fabian Rohner, and Grant J. Aaron. 2017. "Predictors of Anemia in Women of Reproductive Age: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) Project." The American Journal of Clinical Nutrition, June, ajcn143073. doi:10.3945/ajcn.116.143073.
Findings and Implications for the BRINDA Project
Making adjustments for inflammation to iron biomarkers generally increases the prevalence of iron deficiency, and decreases the prevalence of vitamin A deficiency.
Iron deficiency increased by a median of 25 percentage points after applying the regression correction adjustment in children across surveys.
Vitamin A deficiency decreased by a median of 18 percentage points after applying the regression adjustment in children across surveys.
Iron and vitamin A biomarkers need to be adjusted for inflammation.
Overall, the BRINDA project found that inflammation was widespread and there was a relationship between the iron and vitamin A biomarkers with inflammation. Obtaining more accurate estimates of iron (ferritin) and vitamin A (RBP) deficiencies required using inflammation biomarker (AGP and CRP) information to apply an adjustment approach to the iron and vitamin A biomarkers.
The regression correction approach was found to better reflect the relationship between iron and vitamin A with inflammation compared to the correction factor approach. There was a linear relationship between ferritin and RBP with the AGP and CRP biomarkers—meaning that the iron and vitamin A biomarkers were influenced at even low levels of inflammation. Because the correction factor approach only adjusts above clinical thresholds for AGP and CRP, the regression correction approach likely better accounts for the entire effects of inflammation on the iron and vitamin A biomarkers.
Figure 2. Adjustments for Inflammation Make a Big Difference
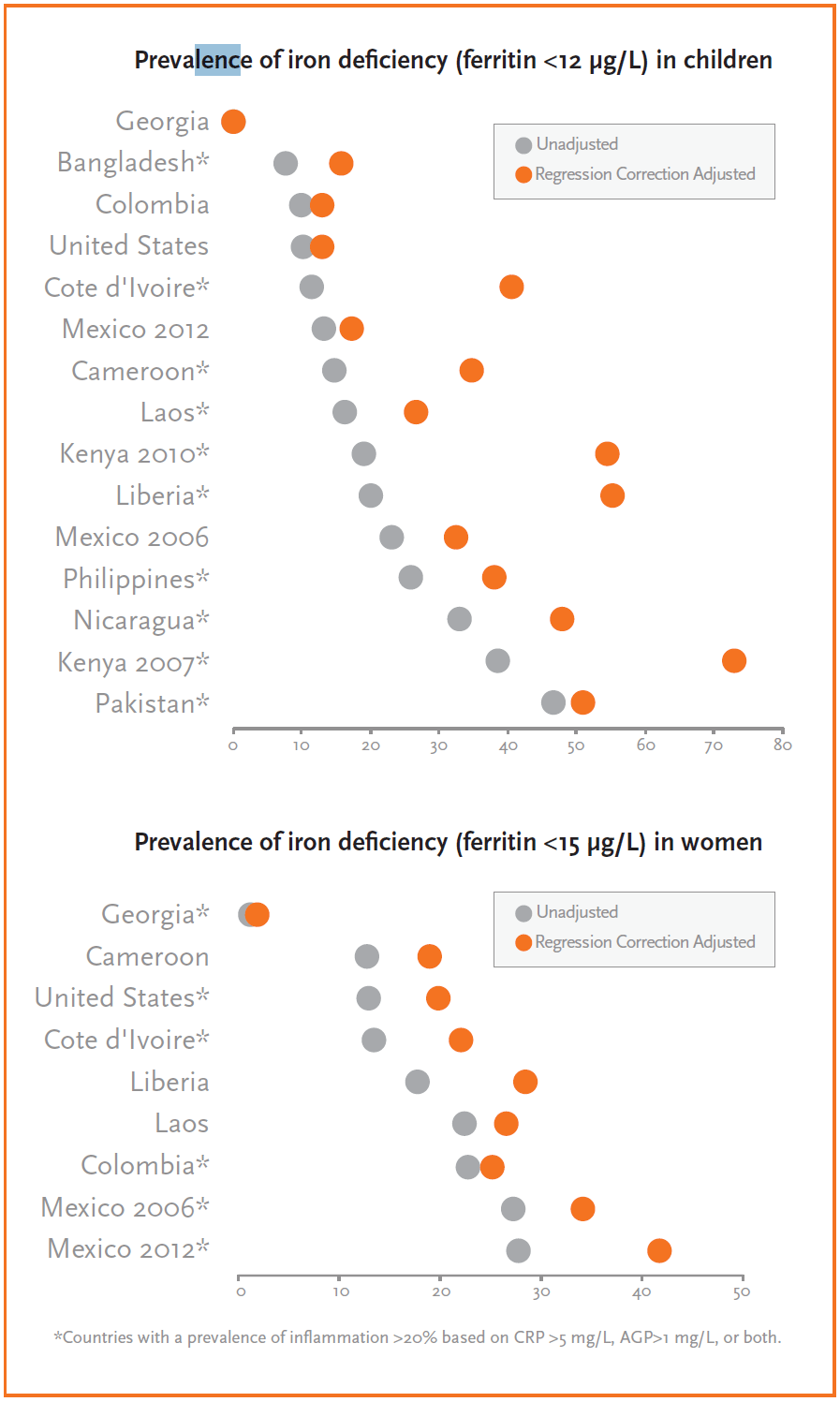
Table 1. Summary of BRINDA Findings on Iron and Vitamin A Biomarker Adjustments for Inflammation and What They Mean for Programs
| BRINDA FINDINGS | PROGRAM IMPLICATIONS |
|---|---|
| The extent to which inflammation exists cannot be determined based on demographic factors or self-reported illness alone. Using AGP and CRP, the median prevalence of inflammation was 57 percent in children and 20 percent in women across surveys. | Inflammation is very common globally and biomarkers are needed to estimate inflammation at the population level. |
| Inflammation prevalence was in general lower when defined by CRP compared to AGP in children, but among women, it varied by survey. The CRP and AGP biomarkers may each reflect different stages or causes of inflammation. | Two biomarkers of inflammation, AGP and CRP, should be assessed to better capture the inflammation situation in a population. |
| Iron and inflammation biomarkers were positively correlated—as AGP and CRP values increased, ferritin values increased. Vitamin A and inflammation biomarkers were negatively correlated—as AGP and CRP values increased, RBP values decreased. | Inflammation biomarkers (AGP and CRP) need to be assessed in conjunction with iron (ferritin) and vitamin A (RBP) biomarkers. |
| Even inflammation below clinically defined thresholds influenced the iron and vitamin A biomarkers. The regression correction approach adjusts for these lower levels of inflammation while the correction factor approach does not. | The regression correction approach should be used when calculating iron and vitamin A deficiencies. Software exists to do so.d |
| Applying inflammation adjustments resulted in a large difference between the unadjusted and adjusted estimates of iron and vitamin A deficiencies, with the regression correction approach resulting in the greatest difference. | Not adjusting for inflammation will likely result in iron deficiency being underestimated and vitamin A deficiency being overestimated. |
| Target population matters for iron. The relationship between the iron and inflammation biomarkers was stronger in children than in women, resulting in a greater difference in the adjusted versus unadjusted estimates of iron deficiency. | Adjustments to the iron biomarker are especially important for children in areas where inflammation is prevalent, but adjustments are also needed for women. |
| Target population matters for vitamin A. The relationship between the vitamin A and inflammation biomarkers was strong in children, but inconsistent in women. | Vitamin A adjustments are needed in areas where inflammation is prevalent for children, but more research is warranted in the case of women. |
| The adjustment approaches were not compared against a gold standard of iron (e.g., bone marrow iron) or vitamin A (e.g., vitamin A liver reserves) because these biomarkers are invasive and availability of data is scarce. | Until validated, both regression correction-adjusted and unadjusted estimates of iron and vitamin A deficiency should be presented. |
Figure 3. Proportion of Anemic Children with Iron Deficiency before and after Adjusting for Inflammation
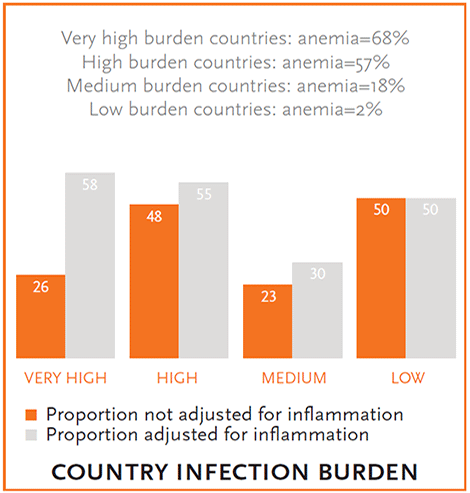
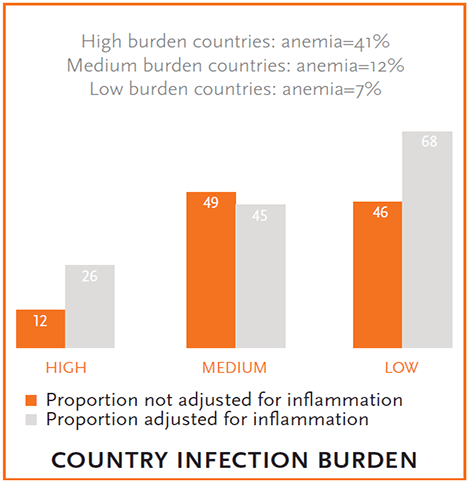
Figure 4. Proportion of Anemic Women with Iron Deficiency before and after Adjusting for Inflammation
Applying the correction factor or regression correction adjustment approach resulted in a higher estimated prevalence of iron deficiency (figure 2) and a lower estimated prevalence of vitamin A deficiency when inflammation was accounted for in countries with a high prevalence of inflammation. The difference was even more pronounced when the regression correction approach was used compared to the correction factor approach. The median percentage point (pp) difference across surveys between the unadjusted iron deficiency prevalence levels and adjusted prevalence levels increased by 25 pp for children and 8 pp for women using the regression correction and by 7 pp for children and 2 pp for women using the correction factor approach. In the case of vitamin A deficiency, there was a decrease of 18 pp and 11 pp for children using the regression correction and correction factor approach, respectively.c A summary of the findings and implications are shown in table 1.
Iron deficiency, inflammation, and infections result in anemia across many settings.
Overall, the BRINDA project found that anemia prevalence varied by survey. The prevalence of anemia ranged from 2 percent (United States) to 84 percent (Bangladesh) among children and 7 percent (United States) to 36 percent (Cameroon and Laos) among women. The prevalence of iron deficiency is high in most countries after making regression correction adjustments to the iron biomarker for inflammation, but there was a wide range of iron deficiency (<1 to 59 percent in children and 1 to 17 percent in women) (figure 2). The proportion of anemic children and women with iron deficiency varied by age group and the level of infectious disease burden in a country (defined by availability of sanitation and safe drinking water, malaria, diarrhea, and schistosomiasis) (figures 3, 4). Countries classified as having the highest burden of infection had the highest burden of anemia (figures 3, 4). In addition, as the infectious disease burden increased, the proportion of anemic children classified as having iron deficiency (measured by ferritin) also increased substantially when iron deficiency estimates were adjusted for inflammation compared to not adjusted (figure 3). The proportion of anemic women with iron deficiency increased the most after adjusting for inflammation in the high and low disease burden countries (figure 4), perhaps indicating some of the drivers of inflammation for women were not infectious in nature.
In children, the risk factors for anemia varied by the level of infectious disease burden in a country, but younger age, iron deficiency, and stunting were consistently associated with anemia. Inflammation and malaria were also associated with anemia in countries with a high burden of infection (table 2). There was limited micronutrient data for children besides iron and vitamin A biomarkers so the relationship between other micronutrients and anemia should be further explored. In women, iron deficiency was consistently associated with anemia, regardless of the burden of infection in the country. Other risk factors for anemia in women were older age, low socioeconomic status, vitamin A insufficiency, and inflammation in some but not all disease burden countries (table 3). There was limited vitamin A, folate, and B12 biomarker data for women so their relationship with anemia should be further explored. A summary of the findings and implications are shown in table 4.
Table 2. Risk Factors for Anemia among Children by Country Infection Burden Table 3. Risk Factors for Anemia among Women by Country Infection Burden
| POTENTIAL RISK FACTORS FOR ANEMIA | VERY HIGH 4 countries | HIGH 4 countries | MEDIUM 3 countries | LOW 2 countries |
|---|---|---|---|---|
| COUNTRIES POOLED BY CLASSIFICATION OF INFECTION BURDEN | ||||
| Age in months (older) | ✓ | ✓ | ✓ | ✓ |
| Inflammation | ✓ | ✓ | ○ | ○ |
| Iron deficiency | ✓ | ✓ | ✓ | ✓ |
| Malaria | ✓ | NA | NA | NA |
| Male child | ✓ | ○ | ○ | ○ |
| Stunted | ✓ | ✓ | ✓ | ✓ |
| Unimproved sanitation | ○ | ✓ | ○ | ○ |
| Vitamin A deficiency | ○ | ○ | ○ | ○ |
| STATISTICALLY SIGNIFICANT: ✓ NOT STATISTICALLY SIGNIFICANT: ○ | ||||
NOTE: Multivariable logistic regression models. Countries classified into infectious disease burden categories using external data collected on a country's sanitation, drinking water, malaria, diarrhea, and schistosomiasis situation. DEFINITIONS: Statistically significant: <0.05. Not statistically significant ≥0.05. Anemia: hemoglobin <110 g/L. Age as a continuous variable in months. Inflammation: CRP >5 mg/L or AGP>1 mg/L. Iron deficiency: inflammation-adjusted ferritin <12 μg/L. Malaria is tested by rapid diagnostic test or microscopy. Stunted height-for-age Z-score <-2 SD. Unimproved sanitation is defined using UNICEF definitions. Vitamin A deficiency: inflammation-adjusted RBP or retinol <0.70 μmol/L. NA: not available. Only presenting potential risk factors with p<.10 in bivariate models, except for micronutrient deficiency and inflammation variables that were forced into the model. | ||||
Table 3. Risk Factors for Anemia among Women by Country Infection Burden
| POTENTIAL RISK FACTORS FOR ANEMIA | HIGH 5 countries | MEDIUM 2 countries | LOW 2 countries |
|---|---|---|---|
| COUNTRIES POOLED BY CLASSIFICATION OF INFECTION BURDEN | |||
| Age in years (older) | ○ | ✓ | ✓ |
| B12 deficiency | ○ | ○ | NA |
| Folate deficiency | ○ | ○ | ○ |
| Inflammation | ✓ | ○ | ✓ |
| Iron deficiency | ✓ | ✓ | ✓ |
| Lower socioeconomic status | NA | ✓ | NA |
| Malaria | ○ | A | NA |
| Vitamin A insufficiency | ✓ | NA | NA |
| STATISTICALLY SIGNIFICANT: ✓ NOT STATISTICALLY SIGNIFICANT: ○ | |||
Note: Multivariable logistic regression models. Countries classified into infectious disease burden categories using external data collected on a country's sanitation, drinking water, malaria, diarrhea, and schistosomiasis situation. DEFINITIONS: Statistically significant: <0.05. Not statistically significant ≥0.05. Anemia: hemoglobin <120g/L. Age: 40-49 vs 15-39 years. B12 deficiency: cobalamin <150 pmol/L. Folate deficiency: folate <10 nmol/L. Inflammation: CRP >5 mg/L or AGP>1 mg/L. Iron deficiency: inflammation-adjusted ferritin <15 μg/L. Malaria: positive by rapid diagnostic test or microscopy. Socioeconomic status: lower and moderate compared to high defined within each survey with the use of appropriate available data. Vitamin A insufficiency: inflammation-adjusted RBP or retinol <1.05 μmol/L. NA: not available. Only presenting potential risk factors with p<.10 in bivariate models, except for micronutrient deficiency and inflammation variables, that were forced into the model. | |||
Table 4: Summary of BRINDA Findings on Risk Factors for Anemia and What They Mean for Programs
| BRINDA FINDINGS | PROGRAM IMPLICATIONS |
|---|---|
| Anemia was high in low- and middle-income countries, especially among children, but varied by country. The highest prevalence of anemia found was 84 percent in children (Bangladesh) and 36 percent in women (Cameroon and Laos). | Anemia is a major public health problem, but anemia assessments are needed to determine the countries with the highest burden. |
| Anemia was highest among countries with a high infectious disease burden. This is indicative of the role inflammation and infection play in causing anemia and also that these countries are characterized by overall poorer health and economic wellbeing. | Efforts are needed to reduce anemia everywhere, but countries with a higher infectious burden should be prioritized. |
| Adjusting iron deficiency for inflammation resulted in more anemic children with iron deficiency in higher infectious disease countries. For women, adjustments were important in both high and low infectious disease countries. | Failure to adjust for inflammation will lead to an underestimate of the extent to which iron is causing anemia for both children and women. |
| Iron deficiency remained the most consistent factor associated with anemia across all infectious disease burden countries after making inflammation adjustments to iron deficiency in both children and women. | Iron is a major driver of anemia in most settings and iron interventions should be prioritized in countries with iron deficiency. |
| Key risk factors for anemia besides iron deficiency in high infection burden countries were malaria and inflammation in children and women. Vitamin A insufficiency was also associated with anemia in women. | Programs need to be designed to address both the nutritional and infectious causes of anemia. |
| Data was unavailable for several potentially important risk factors for anemia (e.g., helminths, human immunodeficiency virus, genetics, zinc deficiency, etc.) and indicators were not always harmonized making comparisons between countries difficult. | Comprehensive surveys with common indicators are needed to assess anemia causes and understand the problem. |
Conclusion
It is important to understand which factor among the many potential factors is causing anemia in a particular context. This requires assessing risk factors for anemia. Demographic and Health Surveys provide information on the prevalence of anemia and coverage estimates of interventions that have the potential to reduce anemia. The Demographic and Health Survey program also supports Malaria Indicators Surveys, (which provide data on malaria prevalence, treatment, and prevention) and Service Provision Assessment Surveys (which provide data on health facility characteristics and services). However, these surveys are not sufficient to identify the range of factors contributing to anemia. There is a need to identify and use diverse data sources that include information on micronutrient deficiencies, malaria, human immunodeficiency virus, helminths, and genetics where such data exists. There is also a need to collect more and better data to inform decision-making.
When identifying the extent to which micronutrient deficiencies contribute to anemia, it is important to use prevalence estimates of iron and vitamin A deficiencies that have been adjusted for inflammation in children. In the case of women, adjustments should also be made to iron biomarkers, but more research is needed to determine whether adjustments should be made to vitamin A biomarkers. Failure to make adjustments will likely lead to an underestimate of iron deficiency and an overestimate of vitamin A deficiency, especially among children in countries where inflammation is prevalent. In turn, this can lead to incorrectly concluding that vitamin A is playing a more important role and iron is playing a less important role in causing anemia. The role of inflammation in confounding the interpretation of other micronutrient biomarkers needs to be assessed in future studies.
It is necessary to match the cause of anemia to the right intervention. If the factors contributing to anemia are not adequately identified, countries will likely end up delivering interventions that do not address the underlying causes of anemia, resulting in limited (if any) reductions in the prevalence of anemia. Increasing the supply of iron may be necessary—if there is a deficiency in this micronutrient—but insufficient to reduce anemia. Interventions to reduce infections and inflammation (e.g. malaria prevention and treatment or water, sanitation, and hygiene efforts) are also critical to reducing anemia in many settings. Other nutritional and non-nutritional causes of anemia need to be further explored.
In cases where there is insufficient data to determine the causes of anemia, it is important to advocate for the collection of better data (e.g., micronutrient surveys) to improve future planning. Targeting the wrong causes of anemia can be especially detrimental for countries with limited resources. A number of tools now exist to support countries to make better use of existing data to understand their anemia situation and prioritize interventions. Understanding Anemia: Guidance for Conducting a Landscape Analysis provides information to policy makers and program implementers on how to gather, understand, and use anemia-related data to develop anemia strategies15; and the District Assessment Tool for Anemia (DATA) assists program managers in strengthening anemia programming at the district level.16 Given the multiple sectors involved in controlling and treating anemia, it is important to involve perspectives from various sectors and stakeholder groups, including government staff, technical assistance providers, and implementers. Use of these guidance tools should include participation by multiple sectors and stakeholders working together to address the causes of anemia.
Footnotes
a Soluble transferrin receptor and total body iron (combination of ferritin and soluble transferrin receptor) were examined as part of the BRINDA project. You can learn more about the use of these biomarkers in the articles by Fabian et al., accessible here, and Mei et al., accessible here.
b For information on how to apply the correction factor or regression correction approach, refer to Namaste et al., accessible here.
c No adjustments were made when estimating vitamin A status for women because the prevalence of vitamin A insufficiency (defined as RBP <1.05 µmol/L ) across surveys was <2% and the relationships between the inflammation biomarkers and RBP values were inconsistent for women.
d A SAS macro to adjust micronutrient biomarkers with the use of the regression correction approach is provided in the online supplemental materials in the article Namaste et al., accessible here.
References
1 Stevens, Gretchen A., Mariel M. Finucane, Luz Maria De-Regil, Christopher J. Paciorek, Seth R. Flaxman, Francesco Branca, Juan Pablo Peña-Rosas, Zulfiqar A. Bhutta, Majid Ezzati, and Nutrition Impact Model Study Group (Anaemia). 2013. "Global, Regional, and National Trends in Haemoglobin Concentration and Prevalence of Total and Severe Anaemia in Children and Pregnant and Non-Pregnant Women for 1995-2011: A Systematic Analysis of Population- Representative Data." The Lancet Global Health 1 (1): e16–25. doi:10.1016/S2214-109X(13)70001-9.
2 International Food Policy Research Institute. 2016. "Global Nutrition Report 2016: From Promise to Impact: Ending Malnutrition by 2030." [PDF, 4.90MB] Washington, DC: International Food Policy Research Institute.
3 Scholz, B. D., R. Gross, W. Schultink, and S. Sastroamidjojo. 1997. "Anaemia Is Associated with Reduced Productivity of Women Workers Even in Less-Physically-Strenuous Tasks." The British Journal of Nutrition 77 (1): 47–57.
4 Rahman, Md Mizanur, Sarah Krull Abe, Md Shafiur Rahman, Mikiko Kanda, Saki Narita, Ver Bilano, Erika Ota, Stuart Gilmour, and Kenji Shibuya. 2016. "Maternal Anemia and Risk of Adverse Birth and Health Outcomes in Low- and Middle-Income Countries: Systematic Review and Meta-Analysis." The American Journal of Clinical Nutrition 103 (2): 495–504. doi:10.3945/ajcn.115.107896.
5 Balarajan, Yarlini, Usha Ramakrishnan, Emre Ozaltin, Anuraj H. Shankar, and S. V. Subramanian. 2011. "Anaemia in Low-Income and Middle-Income Countries." The Lancet 378 (9809): 2123–35. doi:10.1016/S0140-6736(10)62304-5.
6 Thurnham, David I. 2014. "Interactions between Nutrition and Immune Function: Using Inflammation Biomarkers to Interpret Micronutrient Status." Proceedings of the Nutrition Society 73 (01): 1–8. doi:10.1017/S0029665113003662.
7 (WHO) World Health Organization. 2011. "Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity." [PDF, 335KB] WHO/NMH/ NHD/MNM/11.1. Vitamin and Mineral Nutrition Information System. Geneva, Switzerland: World Health Organization (WHO).
8 Raiten, Daniel J., Sorrel Namasté, Bernard Brabin, Gerald Combs, Mary R. L'Abbe, Emorn Wasantwisut, and Ian Darnton-Hill. 2011. "Executive Summary- -Biomarkers of Nutrition for Development: Building a Consensus." The American Journal of Clinical Nutrition 94 (2): 633S – 50S. doi:10.3945/ ajcn.110.008227.
9 WHO. 2014. "C-Reactive Protein Concentrations as a Marker of Inflammation or Infection for Interpreting Biomarkers of Micronutrient Status." WHO/NMH/NHD/EPG/14.7. Vitamin and Mineral Nutrition Information System. Geneva, Switzerland: WHO.
10 ———. 2011. "Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations." [PDF, 447KB] WHO/NMH/NHD/MNM/11.2. Vitamin and Mineral Nutrition Information System. Geneva, Switzerland: WHO.
11 ———. 2011. "Serum Retinol Concentrations for Determining the Prevalence of Vitamin A Deficiency in Populations." [PDF, 828KB] WHO/NMH/NHD/MNM/11.3. Vitamin and Mineral Nutrition Information System. Geneva, Switzerland: WHO.
12 Thurnham, David I., G. P. McCabe, C. A. Northrop- Clewes, and P. Nestel. 2003. "Effects of Subclinical Infection on Plasma Retinol Concentrations and Assessment of Prevalence of Vitamin A Deficiency: Meta-Analysis." The Lancet 362 (9401): 2052–58.
13 Thurnham, David I., Linda D. McCabe, Sumanto Haldar, Frank T. Wieringa, Christine A.. Northrop- Clewes, and George P. McCabe. 2010. "Adjusting Plasma Ferritin Concentrations to Remove the Effects of Subclinical Inflammation in the Assessment of Iron Deficiency: A Meta-Analysis." The American Journal of Clinical Nutrition 92 (3): 546–55. doi:10.3945/ajcn.2010.29284.
14 Petry, Nicolai, Ibironke Olofin, Richard F. Hurrell, Erick Boy, James P. Wirth, Mourad Moursi, Moira Donahue Angel, and Fabian Rohner. 2016. "The Proportion of Anemia Associated with Iron Deficiency in Low, Medium, and High Human Development Index Countries: A Systematic Analysis of National Surveys." Nutrients 8 (11). doi:10.3390/ nu8110693.
15 SPRING. 2017. "Understanding Anemia: Guidance for Conducting a Landscape Analysis." Arlington, VA: Strengthening Partnerships, Results, and Innovations in Nutrition Globally (SPRING) project.
16 SPRING. 2016. "District Assessment Tool for Anemia: Package." Arlington, VA: Strengthening Partnerships, Results, and Innovations in Nutrition Globally (SPRING) project.

